Greater Accuracy.
Highly Precise.
Founded in 2016, Resolve Molecular Diagnostics (RMDx) has been an innovator in genetic testing. RMDx is proud to expand its advanced lab capabilities and services to include the new Elecsys® Anti-SARS-CoV-2 S antibody test. Our dedicated team and instruments ensure that healthcare providers, employers, corporate wellness, and occupational medicine entities have access to the greatest accuracy, highest precision and fastest results for COVID-19 testing.
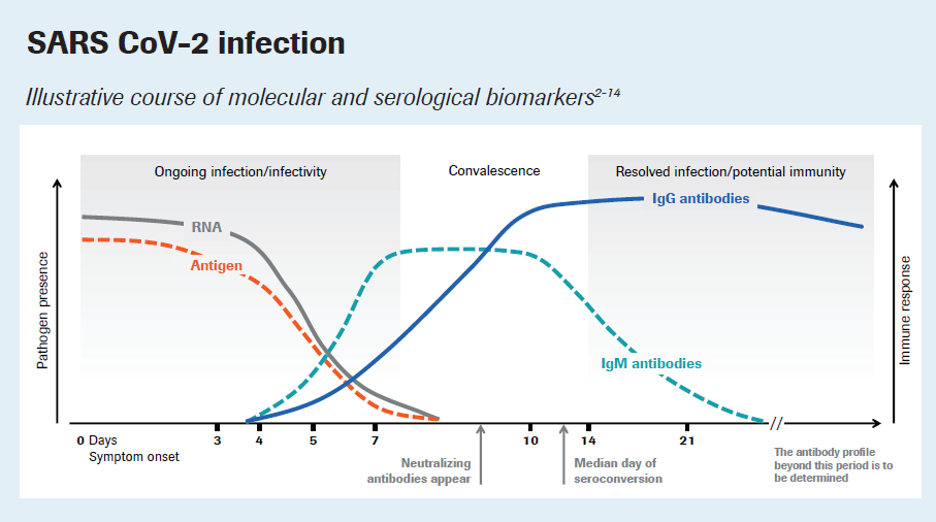
- The Elecsys® Anti-SARS-CoV-2 S is an immunoassay intended for the detection of antibodies to SARS-CoV-2 spike (S) protein in human serum and plasma obtained by a simple blood draw.
- This test can identify individuals who have immunity from prior infection with Covid 19 or by receiving a vaccination.
- Accurate —99.81% specificity achieved through the testing of 5,272 samples and 100% sensitivity ≥ 14 days after PCR confirmation
- Precise —No cross-reactivity found in 80 samples from individuals with past infection to common cold and other coronaviruses
- Scalable — can screen hundreds of thousands of subjects easily.
Elecsys® Anti-SARS-CoV-2 S
NOW AVAILABLE
Covid 19 Spike (S) protein antibodies are triggered to develop via the Moderna, Pfizer, and Johnson and Johnson vaccines. These antibodies are believed to be the “immunity antibodies” against COVID-19.
Resolve Molecular Diagnostics offers the Elecsys® Anti-SARS-CoV-2 S which is an immunoassay intended for the detection of antibodies to SARS-CoV-2 spike (S) protein in human serum and plasma obtained by a simple blood draw. This test can identify individuals who have immunity from prior infection with Covid 19 or receiving a vaccination. The results can be used to confirm immunity or help identify individuals that may or may not need vaccination.
The Elecsys® Anti-SARS-CoV-2
The Elecsys® Anti-SARS-CoV-2 assay is unique among the currently commercially available immunoassays because it detects high-affinity antibodies to SARS-CoV-2. The assay relies on a double antigen sandwich (DAGS) format that enriches detection of higher affinity antibodies, which are more likely to be specific for SARS-CoV-2. Although this assay format is agnostic to the antibody isotype and in principle can detect high affinity antibodies of all isotypes, it preferentially detects IgG antibodies since these are most likely to evolve to become high affinity. The nucleocapsid antigen is abundantly expressed and is a useful target for sensitive detection of virus-specific antibodies. These features provide an optimal combination of high specificity and sensitivity for the detection of immune exposure to SARS-CoV-2.
SUPPORT
When you need support with lab testing, we are here to respond. We serve a variety of practices, providers and patients with personalized customer service and consultative support for lab results. If you have any additional questions, or we can provide any information, please do not hesitate to reach out to our team at 615-800-8471 or support@resolvediagnostics.com.
